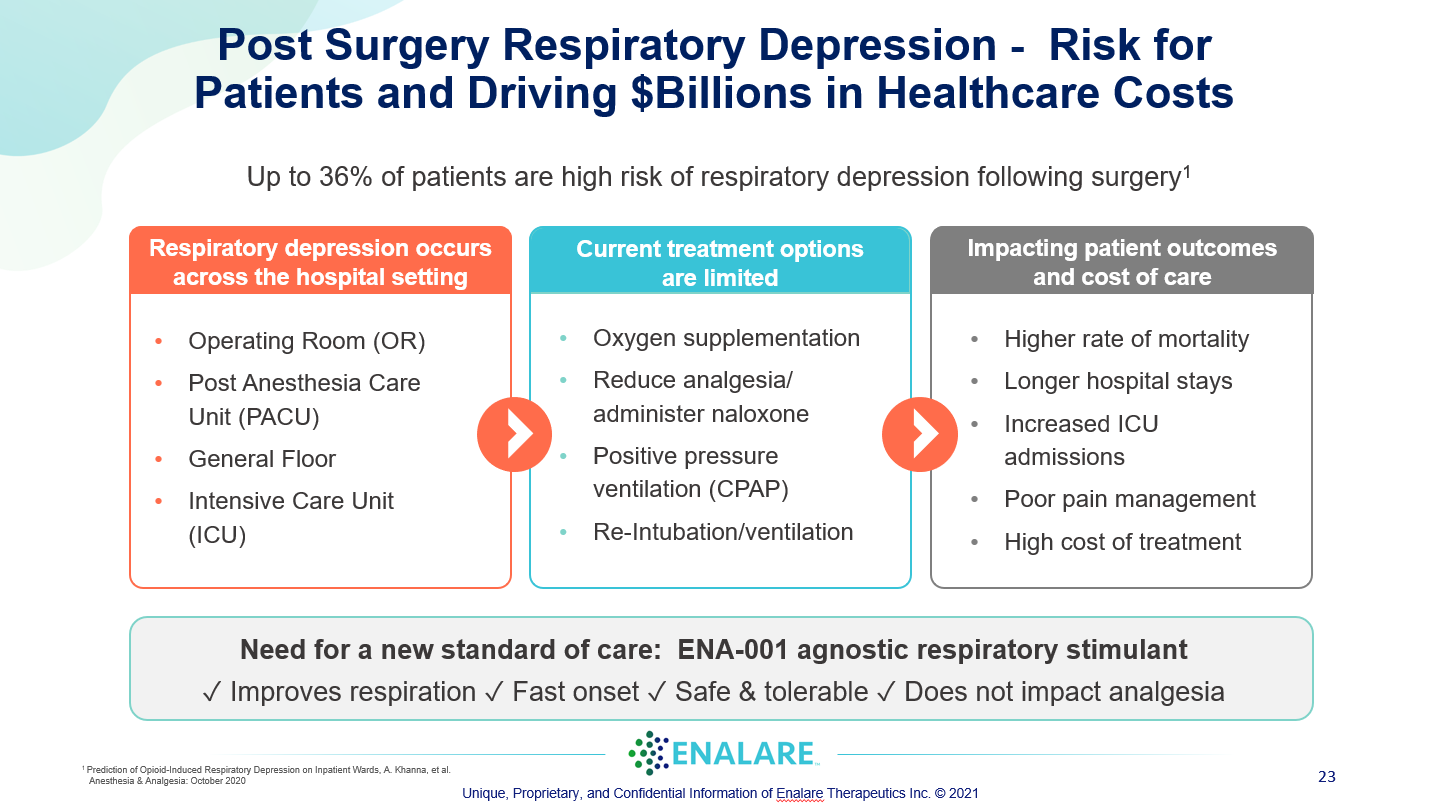
Acute Respiratory Depression is a potentially life-threatening condition characterized by either low level of oxygen (hypoxemia) or high levels of carbon dioxide (hypercapnia). Normal breathing function is a harmonious balance of gas exchange controlled by the central nervous system as well as muscle and peripheral sensory systems. This delicate synchronized mechanism can be disrupted in various manners including use of certain drugs, chronic diseases, and pathogen induced bronchial infections, leading to the dangerous condition of acute respiratory depression.
Enalare Therapeutics
Acute Respiratory Depression
Common Causes of Acute Respiratory Depression
Medications
- Sedative medications
- Narcotic pain medications
- Alcohol
- Other substances that depress brain function
- Combination with benzodiazepines
Health Conditions
- Neuromuscular diseases
- Obesity and aging
- Obstructive sleep apnea
- Chronic lung diseases (e.g. COPD)
- COVID-19 and other pathogens
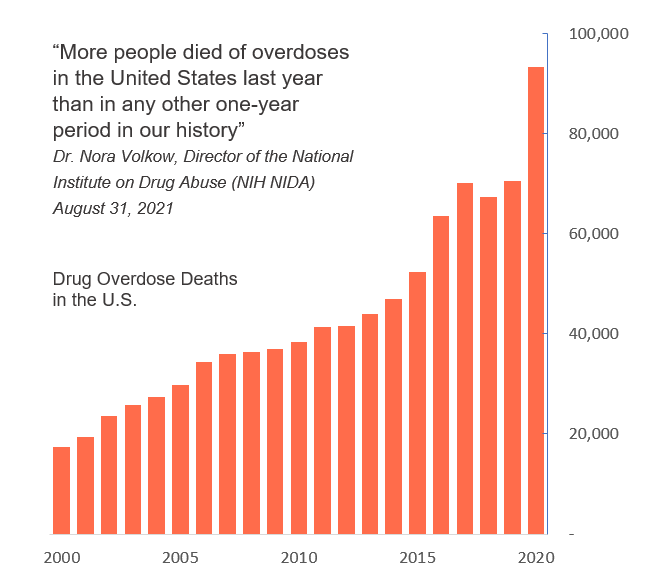
The drug overdose epidemic is escalating and evolving – dire need for an agnostic breathing stimulant
- Drug deaths in the US rose to record levels in 2020
- COVID-19 has only further accelerated this trend, with 95,230 overdose deaths reported by the CDC1
- Poly-pharmacy (multiple drug*) abuse is estimated at greater than 40% and rising2
- For every drug overdose that results in death, there are many more non-fatal overdoses
- Approximately 20mil users misuse opioids and other depressant drugs annually3
- Significant burden on healthcare systems, hospital resources and payors4
- Naloxone (approved in 1971) is the only marketed reversal agent – problematic and incomplete:
- Efficacy limited to opioid overdose
- Agitated patients consume significant ER resources
1 US HHS CDC, 12-month ending Jan. 2021, data available as of Sept. 2021
2 NIH National Institute on Drug Abuse
3 Key Substance Use and Mental Health Indicators in the United States: Results from the 2018 National Survey on Drug Use and Health, SAMHSA, U.S. Department of Health and Human Services
4 Opioid Overdoses Costing U.S. Hospitals an Estimated $11 Billion Annually, Premier Inc, January 2019
Current treatments for drug overdose are inadequate and ill suited for many treatment scenarios
Drug overdose assessment – many unknowns
- Drugs involved, polypharmacy, potency?
- Medical history, concomitant conditions?
- Potential for agitation/ combativeness and other withdrawal symptoms?
Treatment needs:
- Effective agnostic ventilatory stimulant for multiple drug classes
- Fast onset, long duration of action
- No precipitated withdrawal or reversal of analgesia
- Favorable safety profile across patient types
| Approved products | Description | Product Issues |
|---|---|---|
| Naloxone (Narcan) approved 1971 |
An opioid antagonist indicated for the emergency treatment of known or suspected opioid overdose | Opioid withdrawal symptoms Removes pain relief Potential agitation Short duration |
| Flumazenil (Romazicon) approved 1991 | A benzodiazepine receptor antagonist that is indicated for the reversal of conscious sedation induced with benzodiazepines – it can be used as an antidote for benzodiazepine overdose | Contraindications and the possibility of it causing severe adverse effects including seizures, adverse cardiac effects, and death |
| Doxapram (Dopram) approved 1965 | A respiratory stimulant that stimulates chemoreceptors in the carotid bodies. Indicated for drug-induced central nervous system depression and maybe used in patients with mild to moderate respiratory and CNS depression due to drug overdosage |
Side effects include high blood pressure, panic attacks, rapid heart rate, tremor, sweating, and vomiting. Convulsions have been reported. Contraindicated in people with coronary heart disease, epilepsy, and high blood pressure. |
Benzodiazepines: diazepam (Valium), alprazolam (Xanax), clonazepam (Klonopin), etc.
“I just need to make sure the patient is breathing
and then I can focus on treatment”
Emergency Medicine Physician